Understanding whether a precipitate dissolves or not is a fundamental concept in chemistry. One of the most commonly discussed examples in classrooms and laboratories is silver chloride (AgCl). Many students and learners often ask: did the precipitated AgCl dissolve? The answer is not as simple as a yes or no. It depends on the environment, conditions, and chemical interactions involved.
This article explains the concept in a clear and easy-to-understand way, helping you grasp the chemistry behind AgCl precipitation and dissolution.
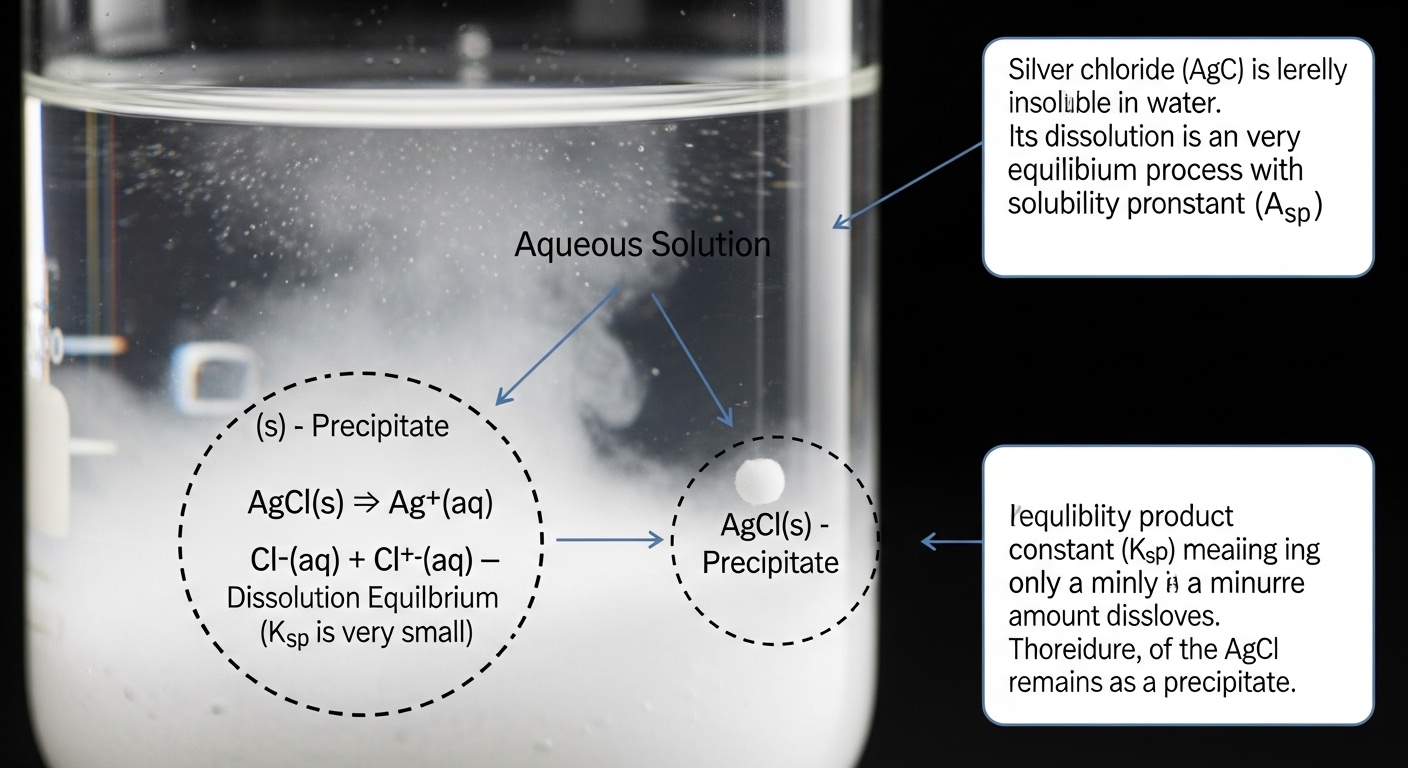
What Is Precipitated AgCl?
To begin, it is important to understand what precipitated AgCl actually is. Silver chloride forms when a solution containing silver ions reacts with a solution containing chloride ions. This reaction produces a white solid known as a precipitate.
For example, when silver nitrate reacts with sodium chloride, silver chloride is formed:
Ag⁺ + Cl⁻ → AgCl (solid)
This solid appears as a white, curdy substance that settles out of the solution. Because it forms a solid from two aqueous solutions, it is called a precipitate.
Did the Precipitated AgCl Dissolve in Water?
When asking did the precipitated AgCl dissolve, the most direct situation to consider is its behavior in pure water. The answer is that AgCl does not dissolve significantly in water. This is because it has very low solubility.
In chemistry, solubility refers to the ability of a substance to dissolve in a solvent. Silver chloride has a very small solubility product constant (Ksp), which means only a tiny amount of it can dissolve in water at equilibrium.
As a result, when AgCl forms, it remains mostly as a solid and does not dissolve back into the solution to any noticeable extent.
Understanding the Solubility Product Concept
To fully explain why AgCl behaves this way, we need to explore the concept of the solubility product, often written as Ksp. This constant represents the equilibrium between the dissolved ions and the solid form of a compound.
For silver chloride, the equilibrium is:
AgCl (solid) ⇌ Ag⁺ (aq) + Cl⁻ (aq)
The Ksp value for AgCl is very small, meaning the reaction strongly favors the solid form. Only a tiny fraction of AgCl dissociates into ions in water.
This explains why, under normal conditions, the precipitate does not dissolve. The equilibrium lies far to the left, keeping AgCl in its solid state.
Did the Precipitated AgCl Dissolve in Ammonia?
While AgCl does not dissolve in water, the situation changes when ammonia is added. This is where the answer to did the precipitated AgCl dissolve becomes more interesting.
When ammonia solution is introduced, it reacts with silver ions to form a complex ion:
Ag⁺ + 2NH₃ → [Ag(NH₃)₂]⁺
This reaction removes free silver ions from the solution. As a result, the equilibrium shifts to the right, causing more AgCl to dissolve in order to replace the lost Ag⁺ ions.
This process continues until much of the AgCl dissolves. Therefore, in the presence of ammonia, the precipitate can dissolve significantly.
The Role of Complex Ion Formation
The dissolution of AgCl in ammonia is an example of complex ion formation. A complex ion forms when a metal ion binds with molecules or ions called ligands.
In this case, ammonia acts as a ligand and binds to silver ions. This stabilizes the dissolved form of silver and reduces the concentration of free silver ions in the solution.
Because of this, the solid AgCl continues to dissolve to maintain equilibrium. This is a key concept in chemistry that explains why some insoluble compounds can dissolve under specific conditions.
Did the Precipitated AgCl Dissolve in Other Solutions?
Apart from ammonia, there are other conditions where AgCl may dissolve. For example, in solutions containing certain ions or chemicals that can react with silver or chloride ions, dissolution may occur.
However, in most common situations, especially in pure water or neutral solutions, AgCl remains insoluble. Its behavior is consistent with the general rules of solubility, which state that most silver salts are insoluble except for a few exceptions.
This is why silver chloride is often used in laboratory experiments to test for the presence of chloride ions.
Factors That Affect the Dissolution of AgCl
Several factors influence whether AgCl will dissolve or remain as a precipitate. Understanding these factors helps answer the question more completely.
One important factor is the presence of complexing agents like ammonia. These substances can bind with ions and shift the equilibrium, leading to dissolution.
Another factor is temperature. Although temperature can affect solubility, its impact on AgCl is relatively small compared to other compounds.
The concentration of ions in the solution also plays a role. According to Le Chatelier’s principle, changes in concentration can shift the equilibrium and affect the amount of dissolved substance.
Common Ion Effect and AgCl
Another important concept related to this topic is the common ion effect. If a solution already contains either silver ions or chloride ions, the solubility of AgCl decreases.
For example, adding sodium chloride to a solution containing AgCl will increase the concentration of chloride ions. This shifts the equilibrium to the left, making the precipitate even less likely to dissolve.
This effect reinforces the idea that AgCl remains mostly insoluble under normal conditions.
Practical Applications of AgCl Solubility
Understanding whether AgCl dissolves is not just a theoretical question. It has practical applications in chemistry and real-world processes.
In qualitative analysis, AgCl is used to identify chloride ions in a solution. When a white precipitate forms after adding silver nitrate, it indicates the presence of chloride.
Further testing with ammonia can confirm this, as the precipitate dissolves, demonstrating the formation of a complex ion.
This behavior is also important in photographic processes and in certain electrochemical applications.
Did the Precipitated AgCl Dissolve: Final Explanation
So, did the precipitated AgCl dissolve? The complete answer depends on the conditions.
In pure water, the precipitated AgCl does not dissolve significantly due to its very low solubility. It remains as a solid because the equilibrium strongly favors the undissolved form.
However, in the presence of ammonia or other complexing agents, AgCl can dissolve. This happens because the formation of complex ions shifts the equilibrium and allows more of the solid to dissolve.
Therefore, the dissolution of AgCl is conditional and depends on the chemical environment.
Conclusion
The question did the precipitated AgCl dissolve highlights an important concept in chemistry. While silver chloride is generally considered insoluble, its behavior changes under specific conditions.
By understanding solubility, equilibrium, and complex ion formation, we can clearly explain why AgCl does not dissolve in water but can dissolve in ammonia. This makes it an excellent example for learning fundamental chemical principles.
Grasping these concepts not only helps in academic studies but also builds a strong foundation for advanced chemistry topics.